Neurodegenerative Diseases
Professor of Neurology Kevin Talbot on genome editing in neurodegenerative diseases, their genetic causes and ...
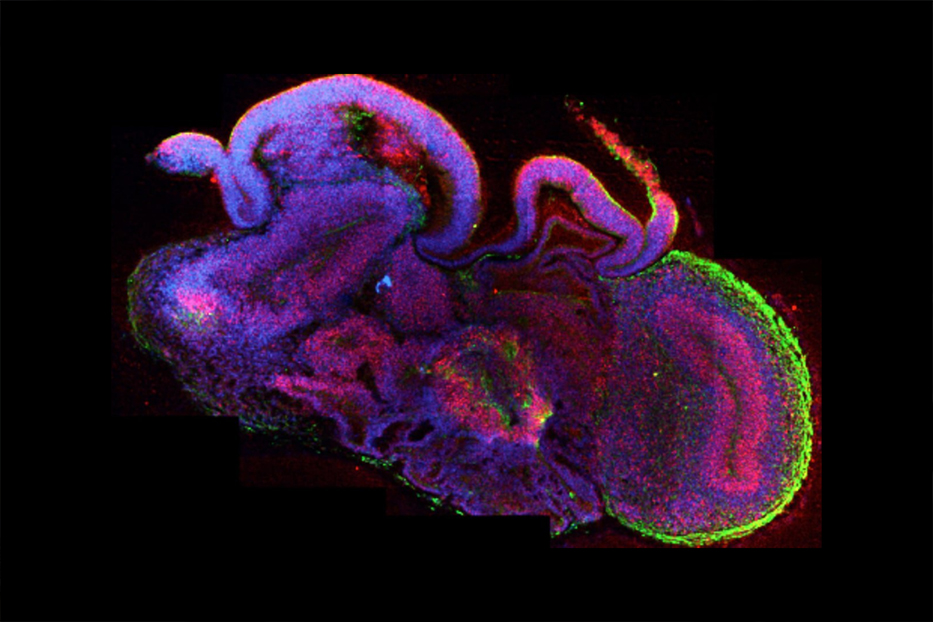
Brain organoids, or cerebral organoids, are three-dimensional miniature brain tissues that develop from stem cells. They grow and form the basic architecture of the developing human brain.
For a long time, neurobiology has relied on animal models, which have been powerful for understanding animal brains. But of course, humans want to understand the human brain. Starting around the beginning of the century, scientists began working with human stem cells, to try and generate human neurons. That has been working well in 2D cell culture, and it is possible to get nice pure populations of specific neuron types. However, these cell cultures are very simple, not really integrated with each other, and have no architecture that you see in an actual human brain. And so that is where I got started.
In order to make a brain organoid, you take stem cells, make a single-cell suspension, and let the cells come back together in an aggregate, forming a ball. And then those cells within that ball start to try to make an embryo, basically. They are obviously disorganized, so they cannot actually make an embryo, but they start to make different germ cell types that are going to generate different organ systems. And then, you push them along towards brain identity so that most of the tissue ends up becoming very primitive brain tissue. The key is – and this comes from the organoid field – putting cells in a specific protein-rich supporting gel, mimicking the surrounding that they would be exposed to in an embryo.
The method that I developed – and there are many methods now since – is very minimalistic. The cells actually know what to do: the brain does develop all by itself in an embryo, without somebody having to go in and add growth factors or other compounds. And there is not actually that much information coming from outside the brain in terms of early patterning: most of it is within that tissue. So all you really need to do is get the cells going by providing the right nutrients to support them. It is a bit like gardening: You plant a seed, and you do not really have to do much. You just give it the right nutrients, sunshine, soil, fertilizer, and water, and you get a plant.
Importantly, we do not want to end up with just a ball of all kinds of different organs. To avoid that, we remove certain growth factors and lipids from the solution in which cells are grown. It is a very simple thing: in the first stages of development, the very first decision that an embryonic or pluripotent stem cell has to make is to decide: am I going to become a brain, or am I going to become everything else? That first decision involves the process of epithelial to mesenchymal transition and gastrulation: the cells that do not undergo gastrulation will become the brain. Those gastrulating cells depend on certain growth factors and lipids, so all you have to do is remove those, and the cells will just make this early, primordial brain tissue.

If you look at our brain organoids, you are not going to see the shape of the brain. Instead, you would see individual brain regions placed next to each other, and each one of those would look the same as a very early-stage individual brain region in a human. So if I took a developing foetal brain and I took apart the brain regions, placed them out on a table, and then would do the same for my brain organoids, you could see that they look similar. It is just the way they are put together in an organoid is almost random: there is still some local patterning so that the brain regions that are supposed to be next to each other tend to be next to each other, but overall it does not have the shape of the brain.
Our brain organoids contain about a million cells and grow up to about half a centimeter in length. So, on the one hand, they are pretty big; you do not need a microscope to see them. On the other hand, for a brain, half a centimeter is very small.
Our organoids are a model of the developing brain, and they cannot develop further and form full-sized brain tissue because they have no blood vessels.
In fact, the human brain develops without blood vessels for the first trimester of pregnancy. At that stage, it has a very thin epithelium with a large fluid-filled cavity on the inside, and blood comes from fetal vasculature on the outside, allowing nutrients to get in by diffusion. That is why our organoids develop so nicely all the way until about mid-gestation because even in development, there would not be blood vessels within the actual brain tissue itself until about the second trimester. Of course, the first-trimester foetal brain is still much bigger than a million cells: the thickness of the epithelium is the same in the organoids and the foetal brain. However, the latter is organized better, so the epithelium there is more continuous, and the fluid-filled space is bigger.
The organoids are making neurons up to about six months: that timing matches in vivo really nicely. While they are making new neurons, the earlier-born neurons are starting to die, but they are being replaced with new neurons, so the organoids continue to grow. However, after six months, the organoids switch and start making glia because that is what they do in vivo as well, and so when the neural stem cells stop making neurons, there is nothing to replace all the cells that are dying. Thus, after about 6 months, when they get to a size of about half a centimeter, they stop growing and start to shrink, and eventually, you end up with a ball of glia, because glia are incredibly robust, and neurons are not.
To overcome this, we developed a special method in our lab: at about the second month, after the organoids have set up the initial architecture, but the neural stem cells are still making neurons, we slice them into thick slices and put them on the air-liquid interface. That way, we can keep the neurons alive for years; we actually have 2.5 years old organoids! It is our way of dealing with the nutrient and oxygen diffusion problem. Of course, these slices are a bit artificial, but that is how we can see beautiful neural tracts forming.
There is a lot of interest in finding a way to incorporate blood vessels into the organoids and, therefore also, overcome the size limitation.
A lot of people think that the initial step that we made, of just developing the organoids in the first place, was a really big jump. I would, of course, like to think so because I had a major part in that, but I actually think it is much harder to get blood vessels in there and get it to develop beyond where it currently is.
So far, from the published studies, it seems that the blood vessels can invade the organoids, but they invade in such a way that the brain tissue is damaged, and that, of course, does not happen in vivo. I think further progress is going to require some careful descriptive studies of what happens in vivo and only then trying to replicate that in vitro.
Modeling neurodevelopmental disease in the organoids is where most of the groups in the field are going now. You could name a neurological disease, and somebody is trying to model it in brain organoids. Probably the most ground will be made on the more severe neurodevelopmental disorders: if you can see something morphologically different in the patients’ brains, then you can probably see something in the organoids.
The first example of a neurodevelopmental disease that was modeled was the genetic form of microcephaly, a condition in which the brain does not develop properly, resulting in a smaller-than-normal head. We could see that the organoids that had the microcephaly mutation were smaller than normal ones. It was a successful proof-of-principle because mouse models for microcephaly are not good: you make the same genetic mutation in a mouse, and the brain looks relatively fine; it is very hard to distinguish it from normal mouse brains. But in human organoids, we could see the size difference and then do cell biology on them and look at what was going on. The underlying problem turned out to be premature differentiation: neural stem cells were making neurons too soon, and so the stem cell population was getting depleted too early.
Since that study, the next logical step was to study the microcephaly that is caused by the Zika virus. If we could model the genetic version of the disease, then could we also model the viral-induced? And that is a big success story of brain organoids. WHO declared Zika a Public Health Emergency in January-February 2016, and by March, the first brain organoid Zika paper was published. Since then, there have been a lot more in-depth studies on what is the Zika virus actually doing, what cells it is targeting, what is receptor it is going for, and why the cells are dying. There is clearly a particular sensitivity of foetal neural stem cells, whereas the adult brain seems fine if it is infected with Zika. So by understanding why foetal stem cells are different, you could understand the mechanism of the disease and find potential treatments. And now, there are studies that have started developing drugs in the brain organoids, and they have found effective compounds that seem to inhibit either the ability of Zika to target neural stem cells, or the response of the neural stem cells, to prevent them from dying.
There have also been studies of other genetic conditions: for example, lissencephaly, where the brain is smooth, lacking the usual folds and grooves. Brain organoids do not generally have folds because they are earlier in development. However, scientists can still look at the cellular defect, and they can see that there is a problem with neuronal migration (i.e., how neurons get from the place they were first made to their final position in the brain), which seems to be the cause of the disorder in patients. Those are just a few examples. I think particularly those kinds of genetic malformations can be modeled very well.
So we can see in our organoids the problems related to the brain architecture, but it is harder to model other disorders. For example, schizophrenia: when you look at a brain MRI of a patient, you cannot tell if they have schizophrenia, i.e., there is nothing in the brain structure that we know of that tells this diagnosis. At the end of the day, what makes a person schizophrenic are behavioral and cognitive difficulties. In organoids, we can look at cell biology, architecture, and molecular identity, but we are barely getting into functional studies, so going all the way to something like schizophrenia or autism is harder.
With autism, there are several forms of the disease that are clearly genetic and monogenic. So there, you can introduce a mutation, for example, in the neurexin gene: neurexin mutation pretty much always leads to autism. The problem is the definition: What do you mean when you say that these patients have autism because it is different for a neurexin patient versus a patient with another mutation? But at least for those cases when you can introduce the mutation, you can also have a control cell line that does not have the mutation, which is very useful. So you can look at cell biological readouts that may inform you about the disease; it can tell you what these genes are doing and why they are needed. Scientists are now doing this kind of work on brain organoids.

And even neurodegenerative diseases are now being investigated in organoids: Alzheimer’s, for instance. I am skeptical about how much we can really model those very late-onset disorders, as they are clearly linked with aging. But it might be possible if you are able to induce some kind of aging-like paradigm in organoids or using cells from patients with an inherited form of Alzheimer’s (though that is the minority of Alzheimer’s cases). The latter was actually done in a recent study, and scientists showed that they could get protein aggregates in there, which are one of the main cellular features of neurodegeneration. They could get the two types of aggregates, both plaques and tangles, that you would see in patients. This is quite remarkable, because in mouse models of the disease you never get both; even to get one, the introduction of several genetic mutations is required in 2D cultures that had not been seen either. So organoids are giving us something we could not get with the previously existing models.
How brains evolved and how they are different from other animals’ brains is one of the basic questions, and I cannot think of any model system other than organoids that you could use to study that. At the moment, the few labs that are studying this are just looking: at making organoids from various species and looking at them to see what is different, as well as using a more molecular approach, doing things like RNA-seq, i.e., determining by sequencing what genes are “active” in different cells in an organoid. So you can make the organoids, dissociate them, and identify specific cell types that were in there. If you do this at different time points, then you find that the different mature cell types – the neurons and the glia – are being made a bit slower in the human compared with the chimpanzee.
We see it in organoids, but this is actually expected from previous studies.
The prevailing model of why we have such big brains is based on something called neoteny, which essentially means that our brains take longer to mature. That applies to all levels of neuron development: from neurogenesis, i.e., just making neurons a bit slower, to neuronal maturation. Our neurons are still very immature, even at birth, and they only get maximal maturity after a few years of life.
Besides, there are additional developmental events that happen, such as pruning and network remodeling during the teenage years. Having such a major neurodevelopmental event at around 15 years of age is totally unheard of in any other species.
The idea behind this neoteny model is that by dragging everything out, it gives each of the cell types, or each of the stages, more of a chance to expand. So for the stages of neurogenesis, it gives them more of a chance to make more neurons: if you spend more time on this, you will make more neurons. And if you spend more time maturing the neurons, the neurons will become more impressive in their maturity. In fact, human neurons have a much more complex structure than mouse neurons, and we see that in our organoids as well. The first time we looked at this, I was amazed: the neural networks looked like spider webs; they were so incredibly complex, with huge and intertangled dendritic trees. The mouse neurons, which I was used to looking at before, are much more simple.
So the maturation is different between different species, but it looks like cell types are the same. You do not find a special type of cell that exists only in humans. At least when you compare within apes, it looks like it is more a question of quantity and not quality. I joke that if you had a chimpanzee who had a brain that expanded three times bigger, which is about our size difference, it would probably be sitting here having coffee, talking, inventing smart devices, etc. But that is different between primates and rodents, as there are some things that have popped up in the former that you do not see in the latter. For example, there are certain neurons called von Economo neurons that are just not found in rodents.
Another big part of the brain organoid field is now electrophysiology, i.e., looking at coordinated neural activity in order to understand how the basic neural networks are set up in the brain. These would be genetically encoded pre-programmed networks, so they are not the kind that is involved in learning or memory.
In our organoids, we see particular rules of how this connectivity forms that match the rules that you see in vivo; that is, particular neurons with particular identities project in a very specific way, and it is pre-programmed. It is not because they are learning anything or receiving any sort of input yet, they are just attracted to particular factors being secreted, probably within the tissue, and that is driving their projection patterns. For example, we know that deep-layer neurons project for a much longer range, whereas more superficial-layer neurons project a shorter range. And that is exactly what we see in our organoids: superficial neurons projecting within the organoid, and more deep layer neurons projecting very far and leaving the organoid, shooting out very long distances, several millimeters.
I would also really like to put some organoids in an MRI to see more macroscopic structures. Other scientists have done this for rodents, and our organoids are about as big as a small rodent brain. For us, on a more macro scale, we may be able to pull out particular rules that are the same from organoid to organoid. And then, we can look at whether that is different between different species’ organoids.
I often get a question, especially from the media, about whether our brain organoids could be conscious. Initially, when somebody asked that question, I immediately said “No!”, as it was so obvious to me: I think if you looked at one, you would also not think they are conscious. But actually, it is important to understand why.
We are interested in human brain evolution and what makes our brains special. And it is not just consciousness because there are other animals with consciousness; it is rather higher-order thinking, language abilities, social development, as well as other cognitive abilities. We are interested in what is different about the human brain that gives us those capabilities. Then, from that knowledge, we can also ask: Well, could the brain organoids have any of those features that give us those capabilities?
When you look at the human brain, compared with, for example, a chimpanzee brain or a mouse brain, the first thing you notice is that it is really big. And it is not just brain size to body size, but absolute numbers. And even though, for example, the elephant’s brain is bigger than ours, we have more neurons in the cerebral cortex. The numbers are very hard to really grasp: we are talking 80 to 100 billion overall. If you put that in terms of how many neurons are made every hour during gestation, it is 12 million. A mouse cortex has 4 million.
So a human developing foetus is making 3 entire mouse cortices every hour.
It is an insane amount of neurons that we have.
So, we have calculated that our organoids have around a million neurons: again, on the one hand, it sounds like a lot, but on the other, it is just a fourth of a mouse cortex, and it is many, many orders of magnitude less than what we have. So I think that is the first reason for discounting the possibility that they are conscious: they are just not big enough.

The second is architecture. The reason our brains work the way they do is that they are connected in a specific way, and information is transferred in a directional fashion. You could take all of our neurons, mash them up, and then make a ball of neurons (similarly to how we make organoids from stem cells), but they would not function like a brain because they are not organized enough.
And the third thing. Christoph Koch, a famous professor in the field of consciousness, has been asked about organoids, and he said: “Well, what would they be conscious of?”. They have had no experience at all. Some people have also tried to draw a comparison with locked-in syndrome in humans, a condition in which a patient is aware but cannot move or communicate verbally due to complete paralysis. But these patients have experienced, they have friends and family, they have a rich life, and then they are locked in. And yes, that is suffering. But an organoid does not have any of that, so what would it be conscious of? Nothing.
In the end, our organoids are about the size of a crayfish’s brain. But a crayfish brain is actually better than our organoids because it has the organization, and it’s inside of a body receiving sensory input and interacting with its environment. So while organoids are miles away from being anything close to conscious, they are very powerful for understanding the development and early stages of the disease.
Edited by Estere Seinkmane

Professor of Neurology Kevin Talbot on genome editing in neurodegenerative diseases, their genetic causes and ...

Neuroscientist Paul Allen on schizophrenia, self-regulation of neural activity, and famous voice-hearers

Pharmacologist David Nichols on the psychedelics, hallucinogen persisting perceptual disorder, and therapeutic...